Introduction
Before we move on through the quagmire of the pharmaceutical drug industry, I wanted to mention one other drug that has been added to the arsenal of cholesterol lowering drugs called PCSK9 Inhibitors, and the genetic high cholesterol condition Familial Hypercholesterolemia (FH).
In addition I would like to discuss anti-diabetic drugs, their mechanism of action and their devastating side effects that affect some.
Cholesterol lowering with PCSK9 Inhibitors

Drug companies are not satisfied with Statin drugs lowering your cholesterol low enough, so now they are pushing for you to take a Statin and a PCSK9 inhibitor which together will probably lower cholesterol to such a level that if has never been seen in nature.
What is the problem with these drug companies, Statins work, they do suppress cholesterol production, but it’s the aftermath destruction that patients have to pay the price for something that does not need lowering in the first place.
These PCSK9 inhibitors are expensive at $50/month, but there is a reason why these drugs were created as explained in the next section.
The website Healthline state:
“Adverse events were reported in 69 percent of people taking evolocumab in the clinical trials. Injection-site swelling or rash, limb pain, and fatigue were some of the reported side effects. In the alirocumab trials, adverse events were reported in 81 percent of participants taking the drug. These included injection-site reactions, muscle pain, and eye-related events. Long-term side effects and risks are not yet known“
And Wikipedia continue to say that the side effects were severe enough so:
“Before the infusions, participants received oral corticosteroids, histamine receptor blockers, and acetaminophen to reduce the risk of infusion-related reactions, which by themselves will cause several side effects.“

Fixing a Mutant Gene can also Lower Cholesterol
So what are these new drugs ?.
Actually I found an article in the NCBI database discussing the function and physiology of the PCSK9 gene, dated 2008, however, they begin the article saying “In the past few years..”since it dates back to 2003. The acronym PCSK9 stands for Proprotein Convertase Subtilisin/Kexin type 9 gene.
It is a gene that helps create an enzyme that binds to a receptor for LDL.
The gene actually provides instructions to make a protein to regulate the amount of cholesterol produced by controlling the number of LDL receptors on the cell.
It appears that this GOI (gene of interest) became just that, as a means to ‘fix’ people with a genetic mutation of this gene that causes Hypercholesterolemia..so how many people have this condition???, well you might be surprised; 1 in 500 in general, but in the UK 1 in 120,000.
What researchers discovered was that Statins actually upregulate PCSK9 putting the brakes on their principal mode of action which is to increase LDL receptors in the liver thus diminishing LDL particles in the blood, therefore PCSK9 inhibition should potentiate(increase) the effects of Statins.

Risk of cognitive disorder with the new cholesterol lowering drug
In 2014, due to early trial evidence, the FDA requested drug developers of these inhibitors to assess any Cognitive adverse effects, and in following trails like the OSLER trial (Open Label study of long term evaluation against LDL Cholesterol) reported that out of 1,104 hypercholesterolemia patients who received 420 mg of ‘Evolocumab’ ( Brand name of PCSK9 inhibitor ) for over 1 year, 3 reported amnesia and 5 with memory or mental impairment <1%.
In 2015 an 11 month follow up study to OSLER showed an increase from 4,465 participants reporting delirium,cognitive,and attention disorders, disturbances, dementia and mental impairment.
In the words of the drug developers it is early days yet but clearly a risk exists concerning cognitive disorders.
*If you want to read more about cholesterol lowering drugs I would direct you to a website called THINCS (The International Network of Cholesterol Skeptics)
Familial Hypercholesterolemia (FH)
According to the documented research, FH is a genetic disorder ( Autosomal dominant pattern) that appears in 2 modes Heterozygous ( where only one gene is abnormal), and Homozygous ( both genes are abnormal) that causes High serum cholesterol levels.
Statistically, Heterozygous gene abnormality occurs in 1 in 500 people in most countries and Homozygous gene abnormality afflicts 1 in a million.
Nessa Carey in her book ‘Junk DNA’ reports that in the UK approx 120,000 people ( total population approx 60 million) live with FH while about 1/3 to 1/2 of the FH sufferers end up with Coronary Artery disease by their mid 50s. These figures show that FH and its severity varies.
Furthermore, according to Wikipedia, FH is classified as type 2 Familial Dyslipidemia.
Type 2 refers to LDL receptor dysfunction caused by the abnormality of the gene, that defines the protein(s) necessary to produce the necessary number of LDL receptors to remove LDL particles from the serum, and this gene is called the LDLR gene.
The type 2 FH has 5 classifications where one classification is associated with PCSK9 ( Proprotein Convertase Subtilisin/Kexin type 9), a protein involved in transporting fat molecules ( incl Cholesterol) by binding to LDL particles from extracellular fluid into the cell, reducing LDL particle concentration.
A drug called PCSK9 inhibitors are used to block the PCSK9 gene which causes more LDLRs to be recycled, in other words it blocks the protein production that regulate the amount of cholesterol produced by controlling the number of LDL receptors.
However PCSK9 is responsible for lipoprotein homeostasis, and in clinical trials there was evidence that their use heightened the risk of cognitive disorders.
Another classification is associated with Apolipoprotein ( B in its ApoB100 form) that is involved with binding to the LDL receptor that is reduced by the mutancy since Glutamine is produced instead of Arginine.
At first glance, the choice of a Statin drug in this special FH condition is the perfect choice.
As we explained in Part 2 the rate limiting step in cholesterol synthesis is regulated by the membrane protein enzyme HMG-CoA reductase that catalyses this step in converting HMG-CoA into Mevalonate.
HMG-CoA is sterol sensing in order that it can down regulate HMG-CoA reductase when cholesterol levels are high.
This down regulation of HMG-CoA reductase is achieved by the interaction of Lanosterol (a natural occuring Steroid that Lanosterol synthase that controls cellular cholesterol levels) accumulation in the membrane of the Endoplasmic Reticulum (ER)( An area within the cell responsible for protein processing) that binds with another protein Insig ( Insulin Induced Gene 1 that regulates cholesterol concentration within cells) within the cell.
If there are an inadequate number of LDL receptors being produced to take up the excess serum cholesterol, the next best thing is to limit the amount of cholesterol synthesis being produced in the first place.
The Statin drug works on the basis of molcular mimicry of HMG-CoA where it competes for binding to the HMG-CoA reductase thus inhibiting the actual biological HMG-CoA reductase, and slowing down the mevalonate pathway.
The question now is, if the membrane domain HMG-CoA reductase is sterol sensing, and cholesterol levels are high, would the down regulation of this enzyme not be enough to maintain appropriate cholesterol level ?.
The same cellular feed back circuitry functions if cholesterol levels are low, by increasing the the biosynthesis of cholesterol, HMG-CoA reductase and proteins that increase the uptake of cholesterol from the external environment i.e LDL receptors.
Bergamot Orange a possible natural therapy to treat Dyslipidemia
It would appear that the existing research does not address this question since the bias is toward the use of Statin drugs, but Cardiologist Stephen Sinatra has put together a formulated product that incorporates Bergamonte ( a small citrus fruit that grows in Calabria Italy called a Bergamot orange that tastes more like a lemon and used to flavor Earl Grey tea).
Most citrus fruits contain flavonoid phytonutrients, but the Bergamot is extremely rich in these health promoting substances consisting of O-Glycosides and C-Glycosides ( these occur naturally in flavonoids which tend to feed our gut bacteria in the small and large intestines ).
Examples of O-Glycosides are Neoeriocitrin (Antioxidant that prevents oxidation of LDL Particles), Naringin (Anti-Inflammatory) and Neohesperidin, Brutieridin (Inhibits HMG-CoA Reductase), Melitidin(Inhibits HMG-CoA Reductase), Rhoifolin 40‐O‐glucoside, chrysoeriol 7‐O‐neohesperidoside‐40‐O‐glucoside, diosmin, rhoifolin, chrysoeriol 7‐O‐neohesperidoside, narirutin, and neodiosmin.
C-Glycosides include apigenin 6,8-di-C-glucoside, diometin 6,8-di-C-glucoside, lucenin‐2, vicenin‐2, stellarin‐2, lucenin‐2‐40‐methyl ether, scoparin, and orientin 40‐methyl ether.
Flavonoid C-glycosides significantly show antioxidant activity, anticancer and antitumor activity, hepatoprotective activity, anti-inflammatory activity, anti-diabetes activity, antiviral activity, antibacterial and antifungal activity, and so on.
Most of the time C-glycosyl flavonoids show higher antioxidant and anti-diabetes potential than their corresponding O-glycosyl flavonoids.
Outcomes of Bergamot studies
It is acknowledged that oxidised cholesterol can damage the arterial epithelium, possibly leading to the accumulation of debris in and along the arterial wall producing an atherosclerotic environment compromising blood flow.
Studies such as ‘Antioxidant activity of citrus limonoids, flavonoids, and coumarins’ Yu J et al conducted in 2005, identified the antioxidant power of Naringin, Neoeriocitrin, and Rutin contained in the Bergamot fruit lowering LDL particle oxidation.
Another study ‘Naringin has an antiatherogenic effect with the inhibition of intercellular adhesion molecule-1 in hypercholesterolemic rabbits’ conducted by Choe SC in 2001 compared Naringin and Lovastatin showing a reduction in fatty streak formation and macrophage infiltration in endothelial cells (Endothelium) that line the interior of blood vessels and Naringin, but not Lovastatin, had a hepatoprotective effect ( preventing liver damage).
Injured blood vessels and Neointima formation ( arterial scarring) that occur after angioplasty procedures ( arterial stent implants) have also been shown to be reduced in rats after a 14 day administration of Bergamot oil.
A study was conducted in 2009 by Di Donna et al called ‘Statin-like principles of bergamot fruit (Citrus bergamia): isolation of 3-hydroxymethylglutaryl flavonoid glycosides.’ proposed that Bergamot compounds Neohesperidin and Naringin share similar chemical structure to statins.
A computational modelling study ‘On the inhibitor effects of bergamot juice flavonoids binding to the 3-hydroxy-3-methylglutaryl-CoA reductase (HMGR) enzyme’ conducted by Leopoldini M, et al in 2010 confirmed Bergamots statin-like molecules bind to the HMG-CoA compound.
Another study divided into 3 sets of rats where group1 was fed a normal diet (control group), group 2 was fed a high cholesterol (hypercholesterolemic ). and group 3 the same hypercholesterolemic diet + 1ml of bergamot juice daily, for 30 days.
The third group who received the Bergamot juice ( measured 370 ppm of neoeriocitrin, 520 ppm of naringin, and 310 ppm of neohesperidin) had reduced levels of cholesterol (29.27%), triglycerides (46.12%), and LDL (51.72%) and an increase in HDL (27.61%).
The Atherogenic index of Plasma ( AIP ) is claimed to be able to predict cardiovascular risk by calculating the Log of Triglyceride value/HDL cholesterol value measured in mmol/L or mg/dl.
If the resultant AIP value is <0.11 then risk is low, if it is between 0.11 and 0.21 then the risk is intermediate, and if it is >0.21 then the risk is high.
In this experiment AIP = 1.09 ± 0.10 for group 3 compared to 3.09 ± 0.20 for group 2.
Finally, a randomized double-blind placebo-controlled study ‘Hypoglycemic and Hypolipemic Effects of a New Lecithin Formulation of Bergamot Polyphenolic Fraction’ conducted by Mollace V et al in 2018 involved three randomized groups totalling 20 patients with varying levels of Hyperlipidemia, with average measured values of LDL cholesterol > 120 mg/dl and triglycerides > 175 mg/dl, and serum glucose > 110 mg/dl. in each group, Placebo, Bergamot polyphenolic fraction (BPF) and Bergamot polyphenolic fraction phytosome formulation (BPFP).
The BPF group received 2 * 650mg daily doses of the extract formulation, and the BPFP group received 500g twice daily, both groups for 30 days. The BPF group’s total cholesterol diminished from 262 to 196, LDL cholesterol from 175 to 116, and triglycerides from 252 to 170.
Similar results were seen with the BPFP group total cholesterol from 261 to 198, LDL cholesterol from 174 to 113, and triglycerides from 252 to 173.
Prior to this study in 2015 ‘Bergamot Reduces Plasma Lipids, Atherogenic Small Dense LDL, and Subclinical Atherosclerosis in Subjects with Moderate Hypercholesterolemia: A 6 Months Prospective Study.’ conducted by Toth PP, et al using 80 patients (42 male, 38 female) with moderate Hypocholesterolemia ( 160-190 mg/dl), who received 150mg each per day for 6 months decreased total cholesterol from 255 to 224, LDL cholesterol from 159 to 132, and triglycerides from 159 to 133, and an increase in HDL from 50 to 54.
So what does this all mean ??. Let us revisit conventional medicine’s guidelines :
Recommended levels
Healthy levels of cholesterol don’t vary much for typical adults. Variation of recommnded levels tends to change due to other health conditions and considerations.
Cholesterol levels for adults
- Total cholesterol levels less than 200 milligrams per deciliter (mg/dL) are considered desirable for adults. A reading between 200 and 239 mg/dL is considered borderline high and a reading of 240 mg/dL and above is considered high.
- LDL cholesterol levels should be less than 100 mg/dL. Levels of 100 to 129 mg/dL are acceptable for people with no health issues but may be of more concern for those with heart disease or heart disease risk factors. A reading of 130 to 159 mg/dL is borderline high and 160 to 189 mg/dL is high. A reading of 190 mg/dL or higher is considered very high.
- HDL levels should be kept higher. A reading of less than 40 mg/dL is considered a major risk factor for heart disease. A reading from 41 mg/dL to 59 mg/dL is considered borderline low. The optimal reading for HDL levels is of 60 mg/dL or higher.
Cholesterol levels for children
By comparison, acceptable levels of total cholesterol and LDL cholesterol in children are different
- An acceptable range of total cholesterol for a child is less than 170 mg/dL. Borderline high total cholesterol for a child ranges from 170 to 199 mg/dL. Any reading of total cholesterol over 200 in a child is too high.
- A child’s LDL cholesterol levels should also be lower than an adult’s. The optimal range of LDL cholesterol for a child is less than 110 mg/dL. Borderline high is from 110 to 129 mg/dL while high is over 130 mg/dL.
Conclusive remarks
We are aware now that these recommended guidelines indicated above are questionable, but in the case of FH the body’s natural cholesterol clearance and distribution is unbalanced so we need to exogenously manage it.
Patients with heterozygous FH can have total cholesterol levels in the 350-550 mg/dL range.
Patients with homozygous FH have total cholesterol levels in the 650-1,000 mg/dL range.
At the top end these figures are high so there are 2 alternatives, prescription medicine using Statins and/or PCSK9 Inhibitors, or a more natural approach is using Bergamot orange extract.
If I had this condition I would most definately work with a functional medicine practitioner who woulld more likely want to work with the more natural alternative, but if it is not able to reduce the levels that you may need to consider the synthetic drug alternative.
Using both modalities together is not a good idea
Lowering Blood Sugar
The body is quite capable of managing biological processes including blood sugar levels.
As we explained in the previous article
The secretion of insulin from the Pancreatic Beta cells (Islet cells) are regulated by the existence of glucose in the bloodstream, however the molecular and cellular mechanisms involved in insulin secretion are Islet cell signaling proteins called G Protein coupled receptors (GPCR), that regulate communications within the cells and outside of the cells.
The secretion of insulin is stimulated by glucose (GSIS – Glucose stimulated insulin secretion) that is received by a glucose transporter GLUT2 which in turn activates ATP that changes the Potential difference (PD) of the cell.
This PD depolarizes the cell closing the potassium channels and opening the calcium channels and it is the accumulation of calcium that activates the insulin secretion.
The purpose of the insulin signal is to notify the cells to uptake the glucose in the blood.
What interferes with this process is excessive refined sugar intake and chronic nutrient deficiency (specifically Chromium, Vanadium and Zinc) which requires a simple dietary change and that should be the only treatment advice.
Diabetes Type II occurs when the body accumulates excessive levels of serum glucose that exceeds cellular requirements.
In response the body attempts to lower the serum glucose levels by releasing Insulin hormone to stimulate cellular uptake, which we have just stated have reached their glucose uptake threshold.
What occurs then is a a combination of high serum glucose and high insulin levels floating around in the bloodstream. One of the most effective ways to normalise the situation is to begin Intermittent Fasting using dietary Ketogenic fasting which defines a low carbohydrate, moderate protein and high fat diet
But that’s too simple..it can’t possibly work.

Diabetes is Destroying the Social Fabric of Society including the Next Generation
Civilizations that either do not have access, nor the money to buy from supermarkets and fast food outlets, but live off the land, generally do not suffer from diabetes.
I remember watching a documentary where a family originally from Mexico living in the US, both the husband and wife earning meager salaries, would try to buy some vegetables from the supermarket but because of time constraints they would eat fast food most of the week and feed their teenager son and daughter with the same food.
The main reason that they could not afford real food every day because the husband had raging diabetes, that he was trying to control buying expensive drugs.
So on one hand he was trying to lower his blood glucose, and on the other he was raising it.
The WHO reports that diabetes is the leading cause of death in Mexico affecting 24% of men and 21% of women over the age of 35. Mexican children have the world’s highest rate of childhood obesity, clocking in at more than 28% between the ages of 5 and 9, and 38% between the ages 10-19. The Mexican population is about 130 million.
These are Nov 2011 figures, so what is it now ?. In the US diabetes rates are 29 million people, and in the UK 3.8 million.

Soda Plants are Dehydrating Communities
Swigging sugar packed soda drinks simply fuels the diabetic crisis, and on top of that soda companies set up their bottling plants in various countries and use up precious water to produce their product (typically 1 litre of soda requires 3 litres of water to produce), depriving farmers that need the water to cultivate and irrigate their growing lands.
This dehydration of whole communities occurring in 1999 in North east India in Kaladera where, before the arrival of the soda company’s (Coca- Cola) bottling plant, farmers could easily draw water from 40 foot wells, but in 2003 the farmers had to bore wells 200 feet deep to draw water.
In Plachimada, Southern India the same soda company was kicked out in March 2004 because the village council refused to renew their licence, because it had dehydrated the community and contaminated the water supply.
In another case a soda company had set up bottling operations in a small mexican community, dehydrated it and everybody was drinking soda, even the small infants were being bottle fed with coca-cola.

Rosiglitazone the anti-diabetic drug that kills
The infamous anti-diabetic drug that was introduced in 1999 was Rosiglitazone, at the same time when Pioglitazone was around which was another Thiazolidinedione class of drug.
The result of a drug trial conducted in 1999 by its maker SmithKline Beecham (Glaxo) found that the drug had a nasty side effect in some...Heart failure…whoopee.
The blood sugar had been lowered but some suffered a heart attack for their troubles..the regulatory agencies should have immediately requested withdrawal of such a dangerous drug..did they?
In 2004 the WHO sent Glaxo a caution about the cardiac events associated with the drug, and after a meta analysis (extrapolating results from a combination of studies) they confirmed it..wait a minute..so the drug must have been approved..yes it was on May 22 1999 the drug was approved, despite the fact that 36 out of 2902 trial participants suffered Ischaemic cardiovascular events (Coronary heart disease).
You must appreciate that, these communications between drug company, the FDA and WHO were unknown to the public..why would they need to know? They just risk their lives taking the stuff.

Approving a Potential Killer
It was also discovered that this drug also increased LDL by 19%, but this was dismissed saying that the LDL particles were big and fluffy and posed no risk..well the Statinator is going to say something about this.
As the years passed, review articles began voicing their concerns about the cardiovascular association with Rosiglitazone, reports of water retention and Congestive heart failure (CHF) began to surface within the first 2 years of drug launch.
But the FDA did their job with a fake fix and modified the drug packet labelling to reflect these ‘rare’ risks.
One diabetes expert came forward praising the cardiovascular safety of the drug..what ???..either there is a risk or there isn’t..which is it?
It turns out that our physician, the diabetes expert was paid a visit from a delegation of ‘heavies’ from the makers of Rosiglitazone and told basically to shut his mouth or be sued because of a perceived stock price loss of $4 billion.
As the reports of CHF began accumulating, the FDA ‘came to the rescue’ and put the CHF risks in the warning section of the package inserts..now this is laughable since 95% of drug takers nor the prescribing physician ever read them.
The Truth is Revealed

By 2006 sales of Rosiglitazone were $3.3 billion annually..oh I forgot about the other drug Pioglitazone whose sales were $2.8 billion/year. which by the way reported less CHF events but it was associated with Bladder cancer.
Other studies followed including ADOPT, Avandia (brand name for Rosiglitazone) in daily practice (seeding trial) and DREAM, all of which focused on how good this drug lowered blood glucose..and the CHF…well maybe later, in another trial.
However 75 cardiac events were recorded during the DREAM trial and 55 in the placebo group.
Fortuitously GSK the makers of Rosiglitazone were being sued by the state of New York pertaining to an antidepressant drug and as a result of that lawsuit GSK agreed to establish a ‘clinical trial register’ revealing the results of all clinical trials conducted after December 2000.
The actual results from the trials of Rosiglitazone would now be revealed since 35 of the 42 randomized trials were never published.
The meta analysis carried out by Steven Nissen of the Cleveland Clinic foundation in 2007 uncovered around 30-43% increase of Cardiac events associated with this drug (quite a difference from the 36 out of 2902 reported earlier).
Anti diabetes drugs are supposed to lower the risk of Cardiovascular mortality not increase it, so in 2007 this was shocking news for all, except the drug maker who knew all along but chose to keep it quiet. The editorial of the Lancet writes:
“The story of Rosiglitazone is one of death,greed and corruption…The trust between doctor patient , researcher and participant, or author and editor is undermined when the foundations on which evidence is built are treated with such casual contempt.”
From the book ‘Deadly medicines and Organized crime’ Peter Gotzsche
This is what’s written on the Drugs.com website for the Anti-diabetic Rosiglitazone
“Thiazolidinedione antidiabetics such as rosiglitazone may cause or worsen heart failure in some patients. Tell your doctor if you have a history of heart failure. Rosiglitazone should not be used to treat patients who have heart failure with symptoms, or moderate to severe heart failure. You will be monitored for signs of heart failure when you start rosiglitazone and if your dose increases. Contact your doctor at once if you develop symptoms of heart failure (eg, swelling of the hands, ankles, legs, or feet; shortness of breath; sudden unexplained weight gain). Your doctor may need to stop your medicine or change your dose.”

Rosiglitazone is withdrawn in Europe, but still available in the US
Rosiglitazone was withdrawn in Europe on September 2010, but the FDA decided to keep it approved in the US, and in 2009 began the TIDE trial which was scheduled to finish in 2015.
The purpose of this trial was to compare Rosiglitazone against Pioglitazone in terms of cardiovascular safety, but as we stated before we know the answer and so did the drug maker.
Since the US and Europe refused to supply trial participants GSK went to India..plenty of people there to exploit.
In 2010, the Indian drug regulator halted the trial (2 FDA officers also suggested to stop the trial..wow..talk about closing the barn door after the horse has bolted) because by then all and sundry knew that the drug was causing 500 heart attacks and 300 cases of heart failure every month in the US.
Even after all that had occurred the FDA still refused to withdraw the drug but only to issue a warning:
Rosiglitazone should only be used in patients already being treated with the drug, and in those patients whose blood sugar cannot be controlled with other drugs, and who after consulting with their healthcare professional,do not wish to use Rosiglitazone.
So, if you want to lower your blood sugar what risk do you chose Coronary heart failure or Bladder cancer?

Why is there an association of Congestive heart failure (CHF) and Rosiglitazone?
By all accounts this drug does a wonderful job in treating insulin resistance as well as hypertension which is a positive side effect, but it also increases HDL and increases LDL (but not small particle).
The mechanism of action involves Peroxisome Proliferator Activated Receptors (PPARs).
What the hell are PPARs ?.
These are Nuclear Proteins (protein class that sense steroid and thyroid hormones) receptors that regulate specific gene expression.
Since they can directly bind to DNA and regulate adjacent genes they are classified as transcription factors (to produce mRNA ‘DNA ‘Xerox’ copies).
Activation is initiated by the binding of a Ligand, a substance that forms a complex for a biological purpose, in this case it results in up/down regulation of gene expression, to control the organism, including development and homeostasis.
PPARγ are expressed in all tissues, the heart, muscle, colon, kidney,pancreas and spleen, and activation ligands are generally Prostaglandin E2 (sigalling molecules made from fatty acids and eicosanoids, which are covered in the Metabolic typing series of articles).
Since adipocytes (Fat cells that make up the adipose or fat tissue of the body) are involved in the pathogenesis of insulin resistance (there are obvious associations between obesity and diabetes since they run together symptomatically).
PPARγ are predominant in adipose tissue specifically in the skeletal muscle and these are the targets to re-regulate (fancy word for interfere with..lol) the gene expression.
These Thiazolidinedione antidiabetics stimulate adipocyte differentiation, producing smaller adipocytes that are more insulin sensitive.
So here is the catch, a very serious side effect is that these drugs cause serious fluid retention, found to be as much as 7% in a monotherapy scenario and 15% when combined with insulin.
The fluid retention begins with a peripheral edema (swelling in the lower part of the body caused by excess fluid) that progresses to a pulmonary edema (fluid in the lungs causing breathing problems) and congestive heart failure ( heart weakening, it does not mean the heart stops, but the body becomes congested with excess fluid and the heart has to work harder).
So the question is, what is fluid retention, and what causes it?
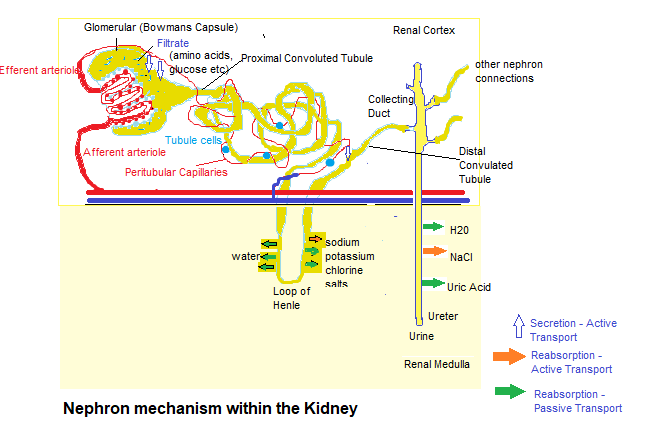
First of all each kidney is made up of about a million filtering units called Nephrons that are made up of a Glomerulus and a tiny tubule, that filter small amounts of blood.
The Glomerulus permits fluid and waste products to pass through it, and block the flow of blood cells and large molecules (mostly proteins).
The filtered fluid then passes through the tubule, sending minerals into the bloodstream and removing waste.
The final product is excreted as urine.
The kidney is also responsible for fluid balance and electrolyte balance of sodium and potassium, so Renal physiology includes Sodium reabsorption.
Evidence suggests that the Thiazolidinedione antidiabetics induces an edema associated with reduced urinary sodium and water excretion directly affecting the Tubular transport and Vascular hyperpermeability, and excessive leakage of fluid and proteins from blood vessels to the interstitial space (space between cells) thus blowing up the affected area or organ.
In other words the drug interferes with the fluid balance responsibility of the kidney.
Two bodies of evidence indicated that, the activation of Sodium transport processes in the Distal Nephron (Convoluted tubule) may underlie the drug-induced fluid retention.
First, within the kidney, PPARγ is highly expressed in the Renal Medullary Collecting Duct, with lower expression levels in Glomeruli, Proximal Tubules, and Microvasculature (smallest system of blood vessels).
Second, in a cultured human Cortical Collecting Duct (CCD) cell line, PPARγ agonists increased levels of cell surface α-ENaC. which are Epithelial Sodium Channels that are involved in the reabsorption of sodium ions at the collecting ducts of the Nephrons.
These channels are critical for the maintenance of body salt and water homeostasis.
In essence the drug interferes with the fluid balance of the kidneys, causing the build up of bodily fluid.
I have included a diagram of the Kidney to give you an idea of the kidney structure:
Conclusion
Can you imagine if Alternative Medicine prescribed natural substances that interfered with human physiology to this extent, it would be like the ‘Blacklist‘ where a special task force would be assembled to shut down every health food store and close every naturopath practice until they were no more.
All alternative practitioners would be on the FBI/Interpol’s most wanted list.
In the next article we’ll look at the regulatory agencies and their role in protecting us citizens….lol.

“According to the surgeon general, obesity today is officially an epidemic; it is arguably the most pressing public health problem we face, costing the healthcare system an estimated $90 billion a year.
Three of every five Americans are overweight; one of every five is obese.
The disease formerly known as adult-onset diabetes has had to be renamed Type II diabetes since it now occurs so frequently in children.
A recent study in the Journal of the American Medical Association predicts that a child born in 2000 has a one-in-three chance of developing diabetes. (An African American child’s chances are two in five.)
Because of diabetes and all the other health problems that accompany obesity, today’s children may turn out to be the first generation of Americans whose life expectancy will actually be shorter than that of their parents.
The problem is not limited to America: The United Nations reported that in 2000 the number of people suffering from overnutrition–a billion–had officially surpassed the number suffering from malnutrition–800 million.”
Michael Pollan, The Omnivore’s Dilemma: A Natural History of Four Meals 2007
Check out the Previous Article in this series:
https://www.extremehealthacademy.com/selling-sickness-part-1-conventional-medicine-is-a-business/
https://www.extremehealthacademy.com/selling-sickness-part-2-profits-before-health/
https://www.extremehealthacademy.com/selling-sickness-part-3-baffling-the-masses/
https://www.extremehealthacademy.com/selling-sickness-part-4-prostaglandin-drugs/
https://www.extremehealthacademy.com/convential-medicine-part-5-the-business-of-antidepressants/
https://www.extremehealthacademy.com/selling-sickness-part-6-cholesterol-lowering-drugs-part-1/
https://www.extremehealthacademy.com/selling-sickness-part-7-cholesterol-lowering-drugs-part-2/
References/Acknowledgments:
- Destroy Chronic pain Dr Russell Schierling Chiropractor 2016
- PCSK9 function and Physiology A.Petersen, L.Fong, S.Young NCBI 2008
- What should my cholesterol level be at my age 2017 Jenna Fletcher Medical News Today
- The rise and fall of rosiglitazone Steven Nissen European Heart journal 2010
- Unravelling the mechanism of action of Thiazolidinediones C.Khan, L.Chen, S.Cohen Journal of clinical Investigation Dec 2000
- Deadly Medicines and Organized crime Book Peter Gotzsche 2013
- Rosiglitazone Drugs.com
- Renal & Vascular mechanism of Thiazolidinediones-Induced fluid retention T.Young, S.Soodvilai NCBI 2008
- Epithelial sodium channel, α-ENaC, renal medullary collecting duct, Distal nephron, Apheresis Wikipedia
- Glomerular Filtration rate (GFR) The National Kidney Foundation
- Mexico diabetes rates contend for world’s worst ZDNET Lauren Villagran Nov 2011
- Coca-Cola Drinking the world dry Waron Want Nov 2007
Author: Eric Malouin